Enamel is the visible outer layer of our teeth and it is made up mostly of minerals. Minerals are the things that make up rocks, so you can think of enamel like a smooth rock, which may be hard, but it can still be damaged. Have you ever known anyone with a kitchen countertop made of marble? Marble is a lot like our teeth. It is a hard rock that can be made into a smooth, shiny counter. However, if you spill something acidic on it, like soda or wine, it will eat away at that smooth shiny surface, leaving a patch that is stained and rough. If that was your tooth enamel, that rough spot could be a nice place for bacteria to make a home and eventually a cavity.
The minerals that make up our enamel are called apatite. Our bodies build apatite crystals into the enamel of our teeth while they are still in our gums. The type of apatite that our body builds is hydroxyapatite. There is another type of apatite that is more resistant to damage, called fluorapatite. It is made by our bodies when fluoride is in our system.
Fluoride is a natural substance. It comes from the element Fluorine (number 9 on the Periodic Table of Elements). Fluorine is actually a dangerous gas, but when a fluorine atom gains an extra electron, it becomes the anion that we call fluoride and can be carried harmlessly in water or incorporate into other minerals. That is exactly how fluoride exists throughout the world. All water sources have some fluoride in them because the water in the ground runs over and through soil, dissolving minerals that contain fluoride. Most of our food contains fluoride because it is made with that water that has fluoride naturally in it. Fluoride can be artificially added to water sources, too. Sometimes fluoride is added to municipal water supplies to protect the oral health of the whole community. You can find out if your water is fluoridated by visiting the links at the bottom of this blog post.
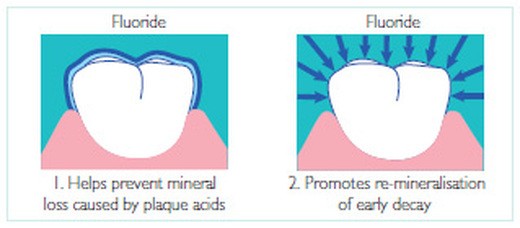
When children drink water with fluoride or take fluoride supplements, their bodies can build stronger enamel on their teeth by making fluorapatite crystals along with the normal hydroxyapatite. In that way, their teeth are stronger right from the start. Fluoride can also help after teeth grow in. Fluoride applied directly to the teeth allows fluorapatite crystals to grow in damaged areas of the enamel. This process is called remineralization. By using fluoride in your toothpaste or mouthwash, you can actually stop and reverse the formation of a cavity. It will only work on little spots of damage, though, so it is best to let your dentist tell you what damage can be fixed with fluoride and what can’t.
-Susan Akers, Blogger & Mom